Procaine Hydrochloride CAS 51-05-8 is the water-soluble hydrochloride salt of procaine. It is the specific formulation used in clinical medicine, famously marketed under the brand name Novocain. While its use in mainstream practice has diminished, it remains a historically pivotal drug and is still employed in specific medical and therapeutic contexts.
Name :
Procaine hydrochlorideCAS No. :
51-05-8MF :
C₁₃H₂₁ClN₂O₂MW :
272.77Purity :
98%Appearance :
White crystalline powderStorage Condition :
Stored in a cool, dry place away from lightChemical Properties
Chemical Name: 2-(Diethylamino)ethyl 4-aminobenzoate hydrochloride
IUPAC Name: 4-Aminobenzoic acid 2-(diethylamino)ethyl ester; hydrochloride
CAS Number: 51-05-8
Molecular Formula: C₁₃H₂₁ClN₂O₂
Molecular Weight: 272.77 g/mol
Chemical Structure: It consists of a procaine molecule (a benzoic acid ester with a terminal diethylamino group) in a salt formation with hydrochloric acid, resulting in a positively charged ammonium ion and a chloride counterion. This salt form is crucial for its solubility and stability in aqueous pharmaceutical preparations.
Appearance: A white, crystalline powder or colorless crystals.
Solubility: Very soluble in water, freely soluble in ethanol. The salt formation dramatically increases its water solubility compared to the procaine free base, making it suitable for injection.
Stability:
Stable in dry form and in acidic solutions (pH ~3.5-5.0 for injections).
The ester bond remains its primary site of instability. It undergoes hydrolysis in neutral or alkaline solutions, accelerated by heat, producing para-aminobenzoic acid (PABA) and diethylaminoethanol.
Solutions should be sterilized by methods that minimize heat exposure (e.g., filtration) and stored protected from light.
pKa: ~8.9 (of the conjugate acid). At physiological pH (7.4), a significant portion exists in the lipid-soluble, uncharged base form, allowing diffusion through nerve sheaths, while the charged cationic form is active at the sodium channel receptor.
Biological Activities
Mechanism of Action: Reversible sodium channel blocker. It diffuses to the neural membrane, and its cationic form binds to receptors within voltage-gated sodium channels, inhibiting sodium ion influx and preventing depolarization and propagation of the action potential, leading to a reversible loss of sensation.
Pharmacokinetics:
Onset: Relatively slow onset of action compared to modern agents like lidocaine.
Duration: Short duration (30-60 minutes for infiltration anesthesia) due to rapid hydrolysis by plasma pseudocholinesterase and significant vasodilation at the site of injection, which increases vascular absorption and removal.
Metabolism: Rapidly hydrolyzed in the plasma by esterases into the metabolites PABA (a known allergen) and diethylaminoethanol.
Toxicity Profile: Lower systemic toxicity than cocaine. Adverse effects (CNS: dizziness, tremors; Cardiovascular: hypotension) are usually associated with high plasma concentrations from intravascular injection or overdose. Allergic reactions, though rare, are primarily linked to sensitivity to PABA or other ester anesthetics.
Biosynthesis
Procaine hydrochloride is not biosynthesized; it is produced via chemical synthesis.
The standard industrial synthesis involves:
1.Esterification: Reaction of 4-aminobenzoic acid (PABA) with ethylene chlorohydrin (2-chloroethanol) to form the ester intermediate, 4-aminobenzoic acid 2-chloroethyl ester.
2.Amination: The chloro-intermediate is then reacted with diethylamine to form procaine free base.
3.Salt Formation: The procaine base is finally treated with hydrochloric acid to precipitate the stable, water-soluble procaine hydrochloride.
Applications

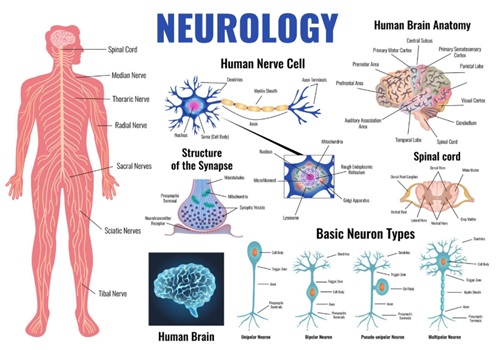

FAQs
Q1: What is the key difference between Procaine (CAS 59-46-1) and Procaine Hydrochloride (CAS 51-05-8)?
A1: Procaine is the free base, a lipophilic compound with limited water solubility. Procaine Hydrochloride is the water-soluble hydrochloride salt of procaine. This salt form is what is used in all injectable pharmaceutical formulations. The CAS numbers are different, confirming they are distinct chemical entities for regulatory and sourcing purposes.
Q2: Is Procaine HCl (Novocain) still commonly used by dentists?
A2: In most developed countries, its use in mainstream dentistry has been largely superseded by amide-type anesthetics like lidocaine, articaine, and mepivacaine. These newer agents have a faster onset, longer duration, and a lower incidence of allergic reactions. It may still be used in specific cases or regions.
Q3: Why is there an allergy warning associated with "caine" drugs, and does it apply to procaine?
A3: True allergy to local anesthetics is rare. However, ester-type anesthetics like procaine are metabolized to PABA. Allergic reactions (like dermatitis or anaphylaxis) are more likely due to sensitivity to PABA or the ester structure itself. Amide-type anesthetics (lidocaine, etc.) do not produce PABA and have no cross-reactivity with esters, making them safer for patients with known ester allergies.
Q4: Why is epinephrine often added to procaine HCl solutions?
A4: Procaine is a vasodilator, increasing local blood flow and causing it to be absorbed into the bloodstream too quickly. Epinephrine, a vasoconstrictor, is added to:
Prolong the anesthetic effect by reducing local blood flow.
Lower the systemic concentration of procaine, reducing the risk of toxicity.
Provide a bloodless field for surgery.
Q5: Can I purchase Procaine Hydrochloride (CAS 51-05-8) for research purposes?
A5: Yes, it is readily available from reputable chemical and biochemical suppliers for legitimate research and development use. Buyers must confirm they are compliant with all local regulations. It is sold as a chemical or pharmaceutical reference standard, not for human or veterinary therapeutic use unless appropriately licensed and formulated.
Q6: What are the main disadvantages that led to its reduced use?
A6: Key limitations include: 1) Short duration of action, 2) Slower onset compared to amides, 3) Potential for allergic reactions (PABA-related), 4) Vasodilatory property, requiring adjuncts like epinephrine, and 5) The development of superior alternatives (amides) with better pharmacokinetic profiles.
Leave A Message
Scan to Wechat/Whatsapp :

